Physician's authorization is required to purchase this item.
Physician's authorization is required to purchase this item.
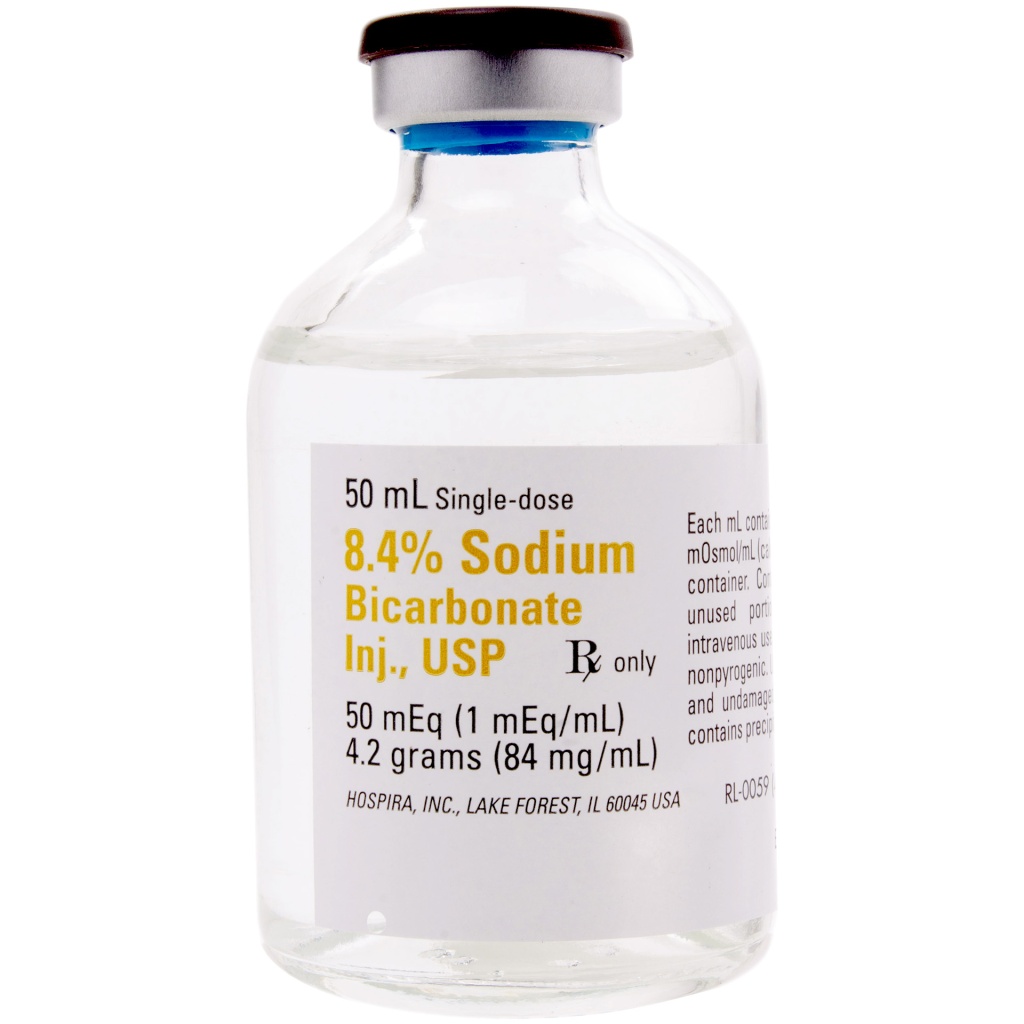
Sodium Bicarbonate Injection, USP is a sterile, nonpyrogenic, hypertonic solution of sodium bicarbonate (NaHCO3) in water for injection for administration by the intravenous route as an electrolyte replenisher and systemic alkalizer.
Solutions are offered in concentrations of 4.2%, 5.0%, 7.5% and 8.4%. See table in HOW SUPPLIED section for contents and characteristics.
Solution in LVP container has 0.9 mg/mL of edetate disodium, anhydrous added as a stabilizer.
The solutions contain no bacteriostat, antimicrobial agent or added buffer and are intended only for use as a single-dose injection. When smaller doses are required, the unused portion should be discarded with the entire unit.
Sodium bicarbonate, 84 mg is equal to one milliequivalent each of Na+ and HCO3¯. Sodium Bicarbonate, USP is chemically designated NaHCO3, a white crystalline powder soluble in water.
Water for Injection, USP is chemically designated H2O.
INDICATIONS AND USAGE
Sodium Bicarbonate Injection, USP is indicated in the treatment of metabolic acidosis which may occur in severe renal disease, uncontrolled diabetes, circulatory insufficiency due to shock or severe dehydration, extracorporeal circulation of blood, cardiac arrest and severe primary lactic acidosis. Sodium bicarbonate is further indicated in the treatment of certain drug intoxications, including barbiturates (where dissociation of the barbiturate-protein complex is desired), in poisoning by salicylates or methyl alcohol and in hemolytic reactions requiring alkalinization of the urine to diminish nephrotoxicity of blood pigments. Sodium bicarbonate also is indicated in severe diarrhea which is often accompanied by a significant loss of bicarbonate.
Treatment of metabolic acidosis should, if possible, be superimposed on measures designed to control the basic cause of the acidosis ― e.g., insulin in uncomplicated diabetes, blood volume restoration in shock. But since an appreciable time interval may elapse before all of the ancillary effects are brought about, bicarbonate therapy is indicated to minimize risks inherent to the acidosis itself.
Vigorous bicarbonate therapy is required in any form of metabolic acidosis where a rapid increase in plasma total CO2 content is crucial ― e.g., cardiac arrest, circulatory insufficiency due to shock or severe dehydration, and in severe primary lactic acidosis or severe diabetic acidosis.